10. Periodic Properties of the Elements
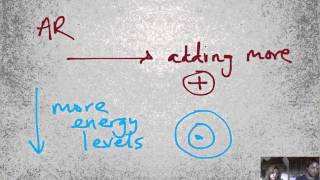
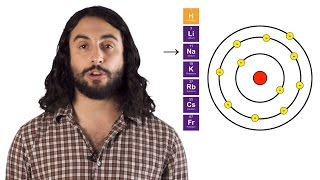
Periodic Trend: Ionic Radius
Multiple Choice
Multiple ChoiceFor an isoelectronic series of ions, the ion that is the smallest is always
A
The ion with the fewest protons.
B
The least positively charged ion.
C
The ion with the highest atomic number.
D
The ion with the most neutrons.
E
The ion with the most electrons.
1860
views
6
rank
Related Videos
Related Practice
Showing 1 of 10 videos