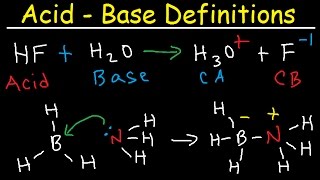
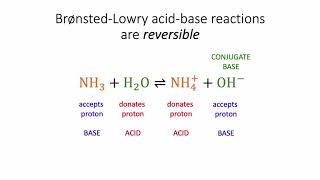17. Acid and Base Equilibrium
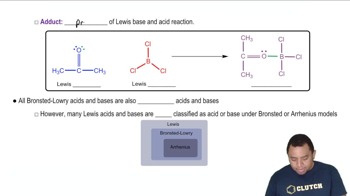
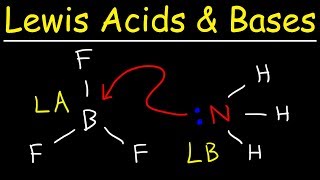
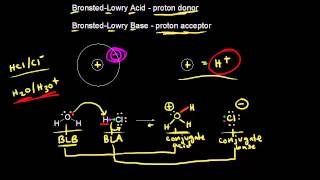
Lewis Acids and Bases
Multiple Choice
Multiple ChoiceIdentify the Lewis acids and bases in the following reactions.
a) H+ + OH– ⇌ H2O
b) Cl– + BCl3 ⇋ BCl4–
c) SO3 + H2O ⇌ H2SO4
A
a) H+:acid, OH-:base; b) Cl-:acid, BCl3:base; c)SO3:acid, H2O:base
B
a) H+:base, OH-:acid; b) Cl-:acid, BCl3:base; c)SO3:base, H2O:acid
C
a) H+:acid, OH-:base; b) Cl-:base, BCl3:acid; c)SO3:acid, H2O:base
D
a) H+:acid, OH-:base; b) Cl-:base, BCl3:acid; c)SO3:base, H2O:acid
3505
views
4
rank
7
comments
Related Videos
Related Practice
Showing 1 of 9 videos