19. Chemical Thermodynamics
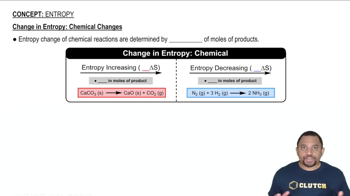
Entropy
Multiple Choice
Multiple ChoiceSelect correct statement(s) below:
a) gaseous CO2 has higher entropy in 2 L container compared to in 5 L container
b) N2O (g) contains higher standard molar entropy then HI (g)
c) NaHCO3 (aq) + HC2H3O2 (aq) → NaC2H3O2 (aq) + H2O (l) + CO2 (g) has a negative ∆S
d) evaporation of water at 100 °C involves greater ∆S than evaporation at 112 °C
A
gaseous CO2 has higher entropy in 2 L container compared to in 5 L container
B
N2O (g) contains higher standard molar entropy then HI (g)
C
NaHCO3 (aq) + HC2H3O2 (aq) → NaC2H3O2 (aq) + H2O (l) + CO2 (g) has a negative ∆S
D
evaporation of water at 100 °C involves greater ∆S than evaporation at 112 °C
272
views
2
rank
1
comments
Related Videos
Related Practice
Showing 1 of 16 videos