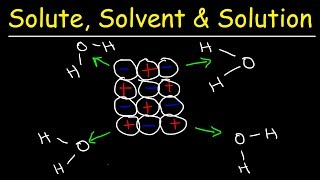
14. Solutions
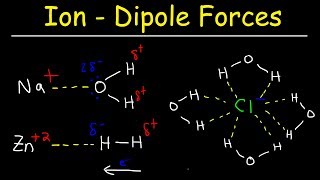
Solutions: Solubility and Intermolecular Forces
Problem 34c
Textbook Question
Textbook QuestionFor each compound, would you expect greater solubility in water or in hexane? Indicate the kinds of intermolecular forces that would occur between the solute and the solvent in which the molecule is most soluble. d. ethylene glycol
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
706
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos