18. Aqueous Equilibrium
Intro to Buffers
Problem 2
Textbook Question
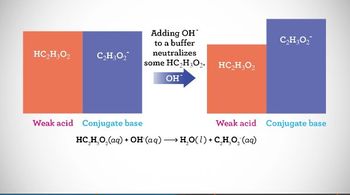
Textbook QuestionThe beaker on the right contains 0.1 M acetic acid solution with methyl orange as an indicator. The beaker on the left contains a mixture of 0.1 M acetic acid and 0.1 M sodium acetate with methyl orange. (b) Which solution is better able to maintain its pH when small amounts of NaOH are added? Explain. [Sections 17.1 and 17.2]

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
636
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos