19. Chemical Thermodynamics
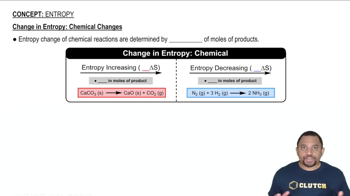
Entropy
Multiple Choice
Multiple ChoiceIdentify sign of entropy changes for the following processes.
1) freezing water to form ice
2) ideal gas allowed to expand in a closed container at constant T
3) mixing of two gases into one container
4) NH2 (g) (1atm) → NH2 (g) (3 atm)
5) gas mixture transferred from larger to smaller container
A
1) −∆S, 2) +∆S, 3) +∆S, 4) −∆S, 5) −∆S
B
1) −∆S, 2) −∆S, 3) +∆S, 4) +∆S, 5) −∆S
C
1) +∆S, 2) −∆S, 3) +∆S, 4) −∆S, 5) +∆S
D
1) +∆S, 2) +∆S, 3) −∆S, 4) +∆S, 5) −∆S
281
views
2
rank
Related Videos
Related Practice
Showing 1 of 16 videos