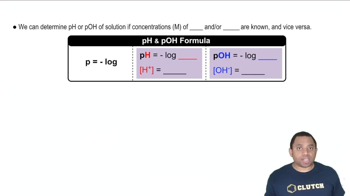
17. Acid and Base Equilibrium
The pH Scale
Multiple Choice
Multiple ChoiceA 345 mL bottle of antacid (Mg(OH)2) contains 1.45 × 10−2 moles of hydroxide ions. Determine pH and pOH of the antacid.
A
pH = 12.384
pOH = 1.616
pOH = 1.616
B
pH = 12.623
pOH = 1.377
pOH = 1.377
C
pH = 12.161
pOH = 1.839
pOH = 1.839
D
pH = 12.338
pOH = 1.662
pOH = 1.662
229
views
5
rank
3
comments
Related Videos
Related Practice
Showing 1 of 10 videos