20. Electrochemistry
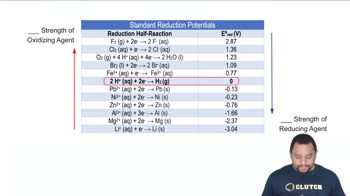
Standard Reduction Potentials
Problem 78
Textbook Question
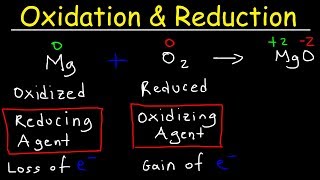
Textbook QuestionConsider the following substances: Fe(s), PbO2(s), H+(aq), Al(s), Ag(s), Cr2O72-(aq). (d) Which substances can be oxidized by Cu2+(aq)? Which can be reduced by H2O2(aq)?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
414
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos