10. Periodic Properties of the Elements
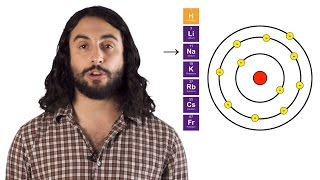
Periodic Trend: Atomic Radius
Problem 88
Textbook Question
Textbook QuestionNote from the following table that there is a significant increase in atomic radius upon moving from Y to La, whereas the radii of Zr to Hf are the same. Suggest an explanation for this effect. Atomic Radii (pm) Sc 170 Ti 160 Y 190 Zr 175 La 207 Hf 175
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
13mPlay a video:
944
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos