13. Liquids, Solids & Intermolecular Forces
Heating and Cooling Curves
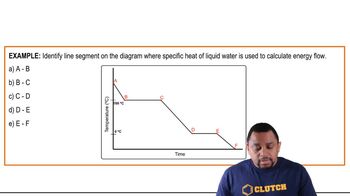
Problem 95
Textbook Question
Textbook QuestionUsing information in Appendices B and C, calculate the minimum grams of propane, C3H8(g), that must be combusted to provide the energy necessary to convert 5.50 kg of ice at -20 °C to liquid water at 75 °C
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
10mPlay a video:
786
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos