17. Acid and Base Equilibrium
pH of Weak Bases
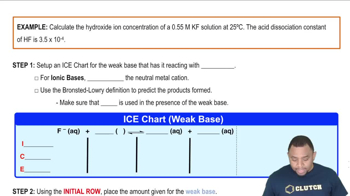
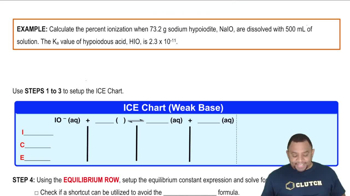
Problem 151
Textbook Question
Textbook QuestionAcid and base behavior can be observed in solvents other than water. One commonly used solvent is dimethyl sulfoxide (DMSO), which can be treated as a monoprotic acid 'HSol.' Just as water can behave either as an acid or a base, so HSol can behave either as a Brønsted–Lowry acid or base. (b) The weak acid HCN has an acid dissociation constant Ka = 1.3 * 10-13 in the solvent HSol. If 0.010 mol of NaCN is dissolved in 1.00 L of HSol, what is the equilibrium concentration of H2Sol + ?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
8mPlay a video:
916
views
Was this helpful?
Related Videos
Related Practice