7. Gases
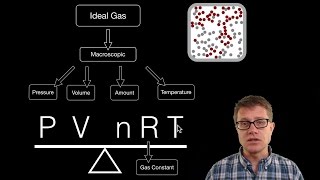
The Ideal Gas Law
Problem 39
Textbook Question
Textbook QuestionA scuba diver's tank contains 2.50 kg of O2 compressed into a volume of 11.0 L. (b) What volume would this oxygen occupy at 25 °C and 101.33 kPa?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
637
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos