18. Aqueous Equilibrium
Ksp: Common Ion Effect
Open Question
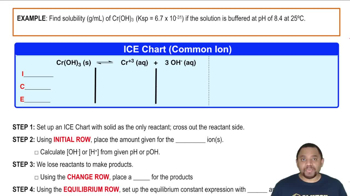
Open QuestionWhat is the solubility of Cr(OH)3 at a pH of 11.10? (Ksp Cr(OH)3 is 6.70 × 10-31)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
96
views
Was this helpful?