12. Molecular Shapes & Valence Bond Theory
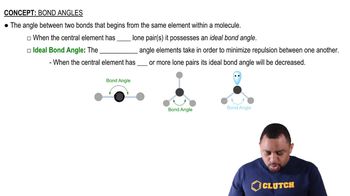
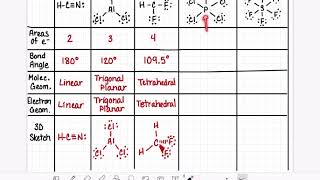
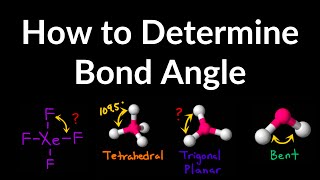
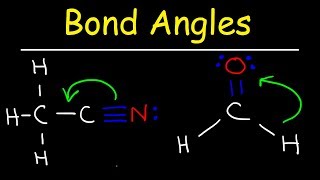
Bond Angles
Problem 31
Textbook Question
Textbook QuestionAmmonia, NH3, reacts with incredibly strong bases to produce the amide ion, NH2-. Ammonia can also react with acids to produce the ammonium ion, NH4+. (a) Which species (amide ion, ammonia, or ammonium ion) has the largest H¬N¬H bond angle? (b) Which species has the smallest H¬N¬H bond angle?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1027
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos