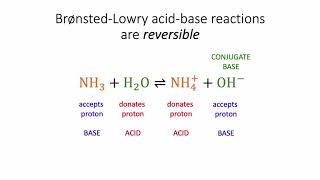17. Acid and Base Equilibrium
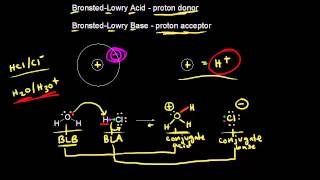
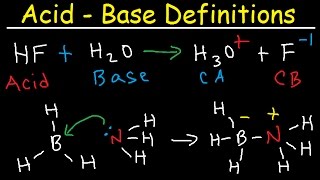
Bronsted-Lowry Acids and Bases
Problem 55
Textbook Question
Textbook QuestionThe hydronium ion H3O+ is the strongest acid that can exist in aqueous solution because stronger acids dissociate by transferring a proton to water. What is the strongest base that can exist in aqueous solution?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
4656
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos