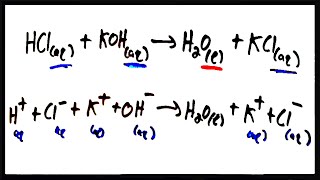6. Chemical Quantities & Aqueous Reactions
Molecular Equations
Problem 38
Textbook Question
Textbook QuestionAssume that an aqueous solution of a cation, represented as a red sphere, is allowed to mix with a solution of an anion, represented as a yellow sphere. Three possible outcomes are represented by boxes (1)–(3):

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
572
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos