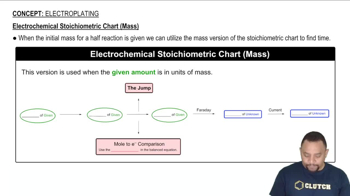
20. Electrochemistry
Electroplating
Problem 151
Textbook Question
Textbook QuestionThe sodium–sulfur battery has molybdenum electrodes with anode and cathode compartments separated by b-alumina, a ceramic through which sodium ions can pass. Because the battery operates at temperatures above 300 °C, all the reactants and products are present in a molten solution. The cell voltage is about 2.0 V. (b) How many kilograms of sodium are consumed when a 25 kW sodium–sulfur battery produces current for 32 min?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
213
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 6 videos