10. Periodic Properties of the Elements
Periodic Trend: Successive Ionization Energies
Problem 78
Textbook Question
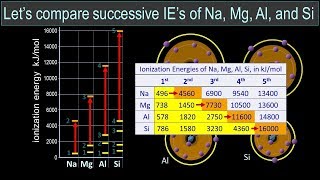
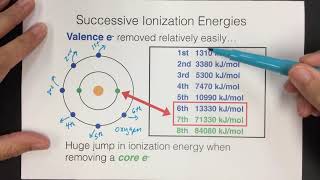
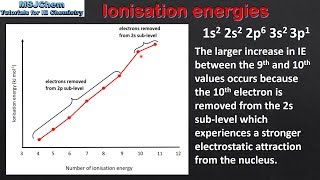
Textbook QuestionConsider this set of ionization energies. IE1 = 578 kJ>mol IE2 = 1820 kJ>mol IE3 = 2750 kJ>mol IE4 = 11,600 kJ>mol To which third-period element do these ionization values belong?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
5773
views
1
rank
1
comments
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos