18. Aqueous Equilibrium
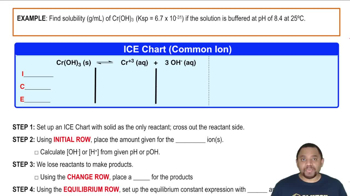
Ksp: Common Ion Effect
Multiple Choice
Multiple ChoiceA solution of Ba(OH)2 has a Ksp of 5.0 x 10−3.
i) Determine the pH of this solution.
ii) Determine the pH if Ba(OH)2 was added to a solution containing 3.2 M of BaF2 and 0.94 M of Al(OH)3.
A
i. 2.30
ii. 14.03
ii. 14.03
B
i. 0.67
ii. 13.55
ii. 13.55
C
i. 13.33
ii. 14.45
ii. 14.45
D
i. 2.30
ii. 13.65
ii. 13.65
190
views
1
comments