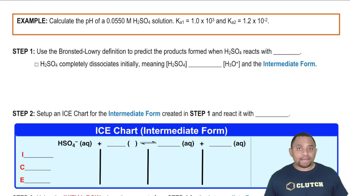
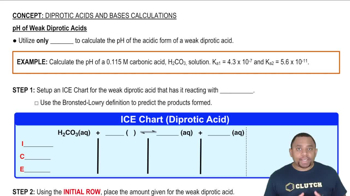
17. Acid and Base Equilibrium
Diprotic Acids and Bases Calculations
Problem 16.116a
Textbook Question
Textbook QuestionConsider a 0.10 M solution of a weak polyprotic acid (H2A) with the possible values of Ka1 and Ka2 given here.
a. Ka1 = 1.0 × 10-4; Ka2 = 5.0 × 10-5
Calculate the contributions to [H3O+] from each ionization step. At what point can the contribution of the second step be neglected?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
16mPlay a video:
95
views
Was this helpful?
Related Videos
Related Practice