18. Aqueous Equilibrium
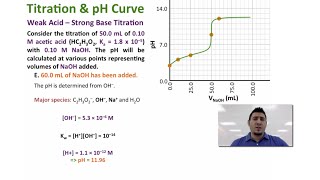
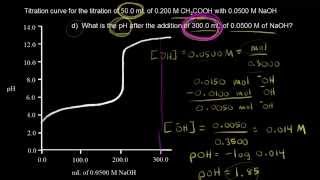
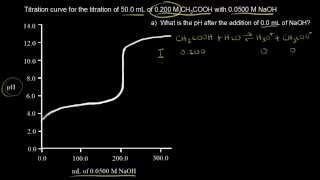
Titrations: Weak Acid-Strong Base
Multiple Choice
Multiple ChoiceCalculate the pH at the equivalence point for the titration of 20.0 mL of 0.500 M HC2H3O2 with 0.350 M KOH. Ka for HC2H3O2 = 1.8 × 10−5.
A
4.99
B
5.63
C
8.37
D
9.01
E
9.25
247
views
Related Videos
Related Practice
Showing 1 of 12 videos