24. Transition Metals and Coordination Compounds
Magnetic Properties of Complex Ions: Octahedral Complexes
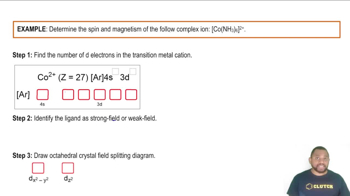
Problem 21.134c
Textbook Question
Textbook QuestionSpinach contains a lot of iron but is not a good source of dietary iron because nearly all the iron is tied up in the oxalate complex [Fe(C2O4)3]3-.
(c) Draw a crystal field energy-level diagram for [Fe(C2O4)3]3-, and predict the number of unpaired electrons. (C2O42- is a weak-field bidentate ligand.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
89
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos