13. Liquids, Solids & Intermolecular Forces
Heating and Cooling Curves
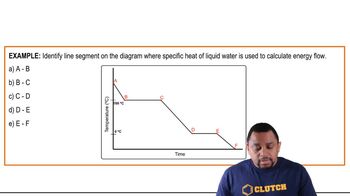
Problem 43
Textbook Question
Textbook QuestionFor many years drinking water has been cooled in hot cli- mates by evaporating it from the surfaces of canvas bags or porous clay pots. How many grams of water can be cooled from 35 to 20 °C by the evaporation of 60 g of water? (The heat of vaporization of water in this temperature range is 2.4 kJ/g. The specific heat of water is 4.18 J/g-K).
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
2108
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos