13. Liquids, Solids & Intermolecular Forces
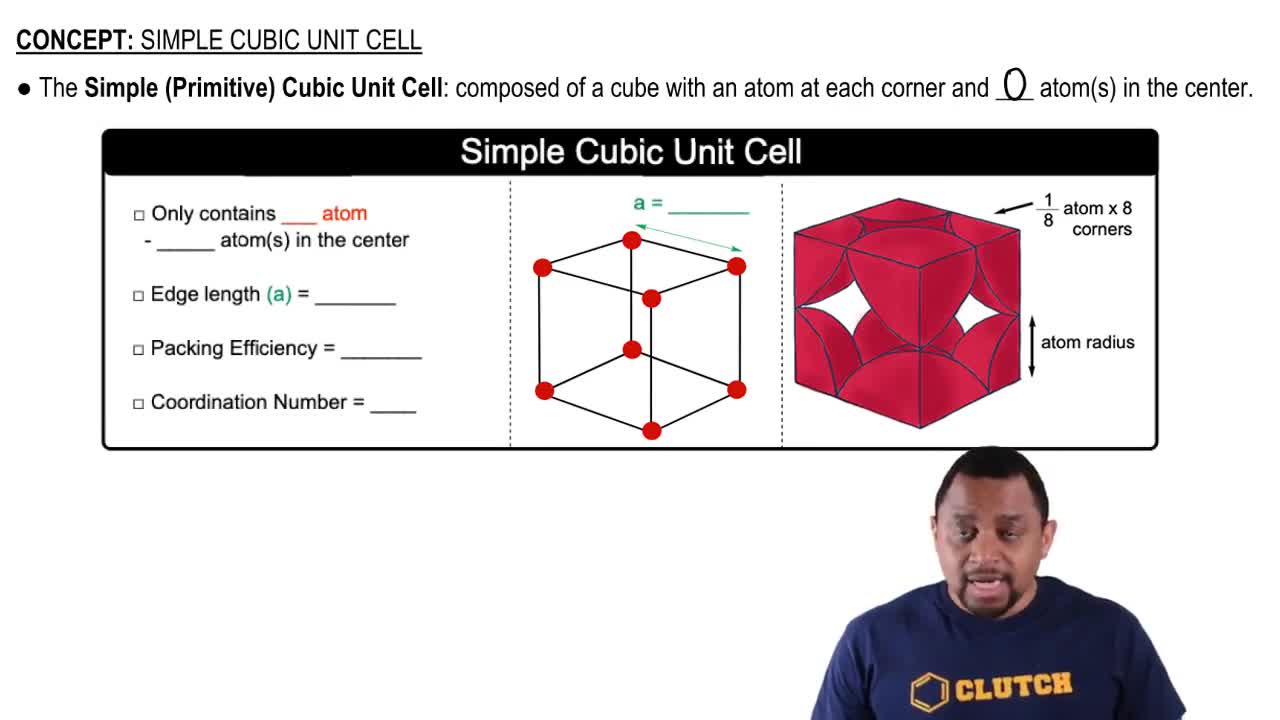
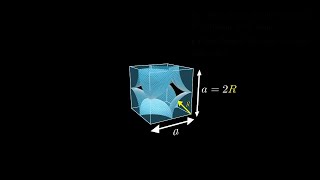
Simple Cubic Unit Cell
Problem 90a
Textbook Question
Textbook Question"The diameter of a rubidium atom is 495 pm We will consider two different ways of placing the atoms on a surface. In arrangement A, all the atoms are lined up with one another to form a square grid. Arrangement B is called a close-packed arrangement because the atoms sit in the 'depressions' formed by the previous row of atoms:

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
15mPlay a video:
789
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos