19. Chemical Thermodynamics
Entropy
Problem 40
Textbook Question
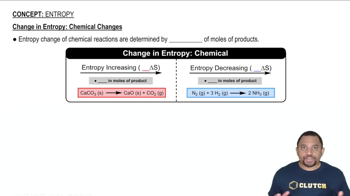
Textbook QuestionFor each of the following pairs, predict which substance possesses the larger entropy per mole: (a) 1 mol of O21g2 at 300 °C, 0.01 atm, or 1 mol of O31g2 at 300 °C, 0.01 atm
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
947
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos