8. Thermochemistry
Enthalpy of Formation
Multiple Choice
Multiple ChoiceConsider the following equation:
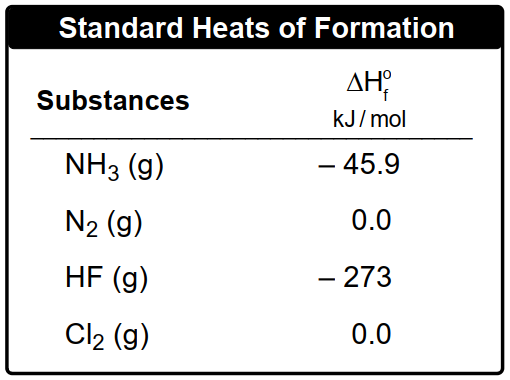
2 ClF3(g) + 2 NH3(g) → 1 N2(g) + 6 HF (g) + 6 Cl2(g) ΔHrxn = –1196 kJ
Determine the standard enthalpy of formation for chlorine trifluoride, ClF3.
A
-175.1 kJ
B
350.2 kJ
C
442.0 kJ
D
-1638 kJ
3083
views
3
comments
Related Videos
Related Practice
Showing 1 of 6 videos