20. Electrochemistry
Electroplating
Problem 162
Textbook Question
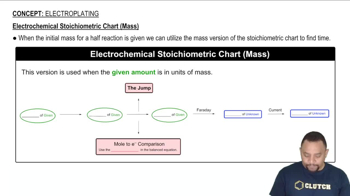
Textbook QuestionThe half-reactions that occur in ordinary alkaline batteries can be written as In 1999, researchers in Israel reported a new type of alkaline battery, called a 'super-iron' battery. This battery uses the same anode reaction as an ordinary alkaline battery but involves the reduction of FeO42- ion (from K2FeO4) to solid Fe(OH)3 at the cathode. (c) A super-iron battery should last longer than an ordinary alkaline battery of the same size and weight because its cathode can provide more charge per unit mass. Quan-titatively compare the number of coulombs of charge released by the reduction of 10.0 g K2FeO4 to Fe(OH)3 with the number of coulombs of charge released by the reduction 10.0 g of MnO2 to MnO(OH).
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
306
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 6 videos