12. Molecular Shapes & Valence Bond Theory
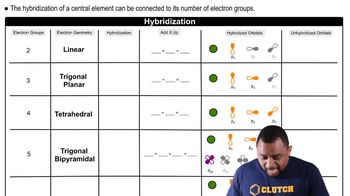
Hybridization
Problem 100
Textbook Question
Textbook QuestionSodium azide is a shock-sensitive compound that releases N2 upon physical impact. The compound is used in automobile airbags. The azide ion is N3-. (b) State the hybridization of the central N atom in the azide ion.
 Verified Solution
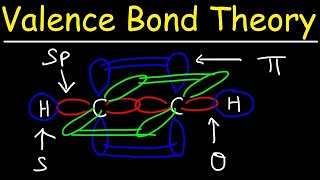
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
716
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos