13. Liquids, Solids & Intermolecular Forces
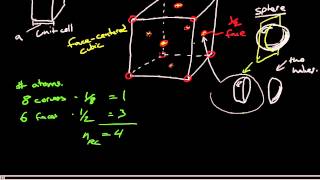
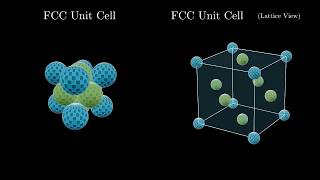
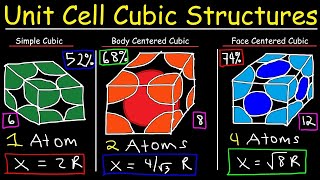
Face Centered Cubic Unit Cell
Problem 78
Textbook Question
Textbook QuestionCopper iodide crystallizes in the zinc blende structure. The sep- aration between nearest neighbor cations and anions is approximately 311 pm, and the melting point is 606 °C. Potassium chloride, by contrast, crystallizes in the rock salt structure. Even though the separation between nearest-neighbor cations and anions is greater (319 pm), the melting point of potassium chlo- ride is higher (776 °C). Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
465
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos