24. Transition Metals and Coordination Compounds
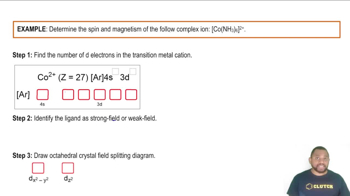
Magnetic Properties of Complex Ions: Octahedral Complexes
Problem 21.137e
Textbook Question
Textbook QuestionThe complete reaction of 2.60 g of chromium metal with 50.00 mL of 1.200 M H2SO4 in the absence of air gave a blue solution and a colorless gas that was collected at 25°C and a pressure of 735 mm Hg. (e) When an excess of KCN is added to the solution, the color changes, and the paramagnetism of the solution
decreases. Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
97
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos