1. Intro to General Chemistry
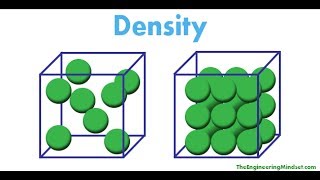
Density
Problem 38a
Textbook Question
Textbook Question(a) After the label fell off a bottle containing a clear liquid believed to be benzene, a chemist measured the density of the liquid to verify its identity. A 25.0-mL portion of the liquid had a mass of 21.95 g. A chemistry handbook lists the density of benzene at 15 C as 0.8787 g>mL. Is the calculated density in agreement with the tabulated value?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
837
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos