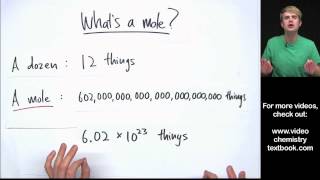2. Atoms & Elements
Mole Concept
Problem 120
Textbook Question
Textbook QuestionAmmonium nitrate, a potential ingredient of terrorist bombs, can be made nonexplosive by addition of diammo-nium hydrogen phosphate, (NH4)2HPO4. Analysis of such a NH4NO3 - (NH4)2HPO4 mixture showed the mass percent of nitrogen to be 30.43%. What is the mass ratio of the two components in the mixture?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
699
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos