24. Transition Metals and Coordination Compounds
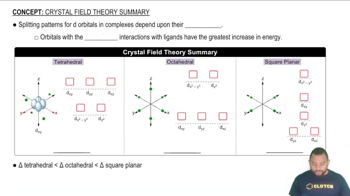
Crystal Field Theory Summary
Problem 21.135a
Textbook Question
Textbook QuestionFormation constants for the ammonia and ethylenediamine complexes of nickel(II) indicate that Ni(en)32+ is much more
stable than Ni(NH3)62+:
(1) <REACTION>
(2) <REACTION>
The enthalpy changes for the two reactions, ΔH°1 and ΔH°2, should be about the same because both complexes have six Ni﹣N bonds.
(a) Which of the two reactions should have the larger entropy change, ΔS°? Explain.
84
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos