19. Chemical Thermodynamics
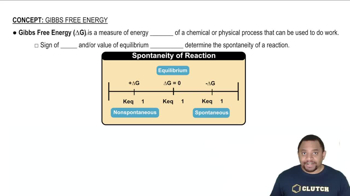
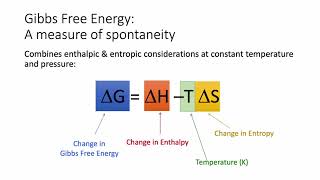
Gibbs Free Energy
Problem 132
Textbook Question
Textbook QuestionTell whether reactions with the following values of ΔH and ΔS are spontaneous or nonspontaneous and whether they are exothermic or endothermic. (a) ΔH = - 48 kJ; ΔS = + 135 J>K at 400 K (b) ΔH = - 48 kJ; ΔS = - 135 J>K at 400 K (c) ΔH = + 48 kJ; ΔS = + 135 J>K at 400 K (d) ΔH = + 48 kJ; ΔS = - 135 J>K at 400 K
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
8mPlay a video:
514
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos