7. Gases
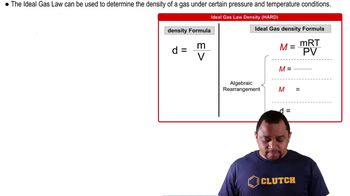
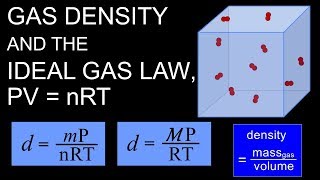
The Ideal Gas Law: Density
Multiple Choice
Multiple ChoiceDetermine the molecular formula of a gaseous compound that is 49.48% carbon, 5.19% hydrogen, 28.85% nitrogen, and 16.48% oxygen. At 27°C, the density of the gas is 1.5535 g/L and it exerts a pressure of 0.0985 atm.
A
C20H44N8O4
B
C16H20N6O2
C
C14H22N2O2
D
C16H20N8O4
758
views
1
rank
2
comments
Related Videos
Related Practice
Showing 1 of 11 videos