18. Aqueous Equilibrium
Intro to Buffers
Problem 4b
Textbook Question
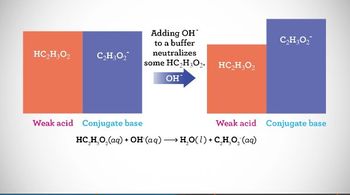
Textbook QuestionThe following diagram represents a buffer composed of equal concentrations of a weak acid, HA, and its conjugate base, A-. The heights of the columns are proportional to the concentrations of the components of the buffer. (a) Which of the three drawings, (1), (2), or (3), represents the buffer after the addition of a strong acid? [Section 17.2]

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
374
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos