13. Liquids, Solids & Intermolecular Forces
Atomic, Ionic, and Molecular Solids
Problem 69a
Textbook Question
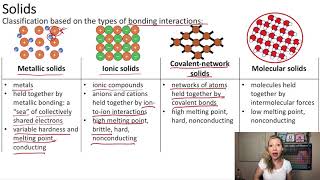
Textbook QuestionBoth covalent-network solids and ionic solids can have melting points well in excess of room temperature, and both can be poor conductors of electricity in their pure form. However, in other ways their properties are quite different. (a) Which type of solid is more likely to dissolve in water?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1148
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos