10. Periodic Properties of the Elements
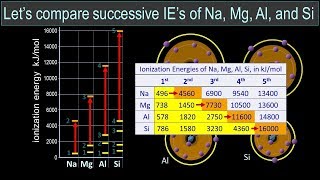
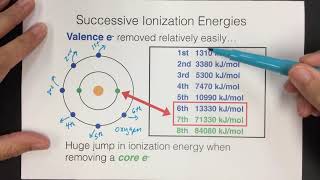
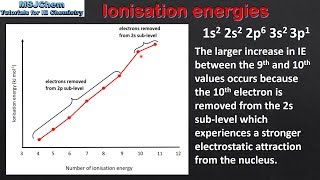
Periodic Trend: Successive Ionization Energies
Multiple Choice
Multiple ChoiceWhich of the following represents the third ionization of Mn?
A
Mn− (g) + e− → 2 Mn2− (g)
B
Mn2+ (g) → Mn3+ (g) + e−
C
Mn2− (g) + e− → 2 Mn3− (g)
D
Mn (g) → Mn3+ (g) + 3 e−
570
views
2
rank
Related Videos
Related Practice
Showing 1 of 10 videos