7. Gases
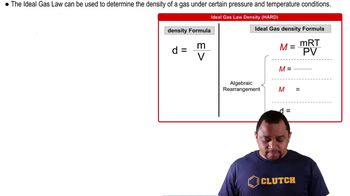
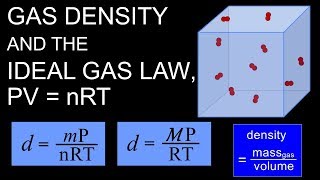
The Ideal Gas Law: Density
Multiple Choice
Multiple ChoiceConsider two containers of gases at the same temperature. One has helium at a pressure of 1.00 atm. The other contains carbon dioxide with the same density as the helium gas. What is the pressure of the carbon dioxide gas sample?
A
0.063 atm
B
0.091 atm
C
0.133 atm
D
1.51 atm
E
2.71 atm
528
views
Related Videos
Related Practice
Showing 1 of 11 videos