13. Liquids, Solids & Intermolecular Forces
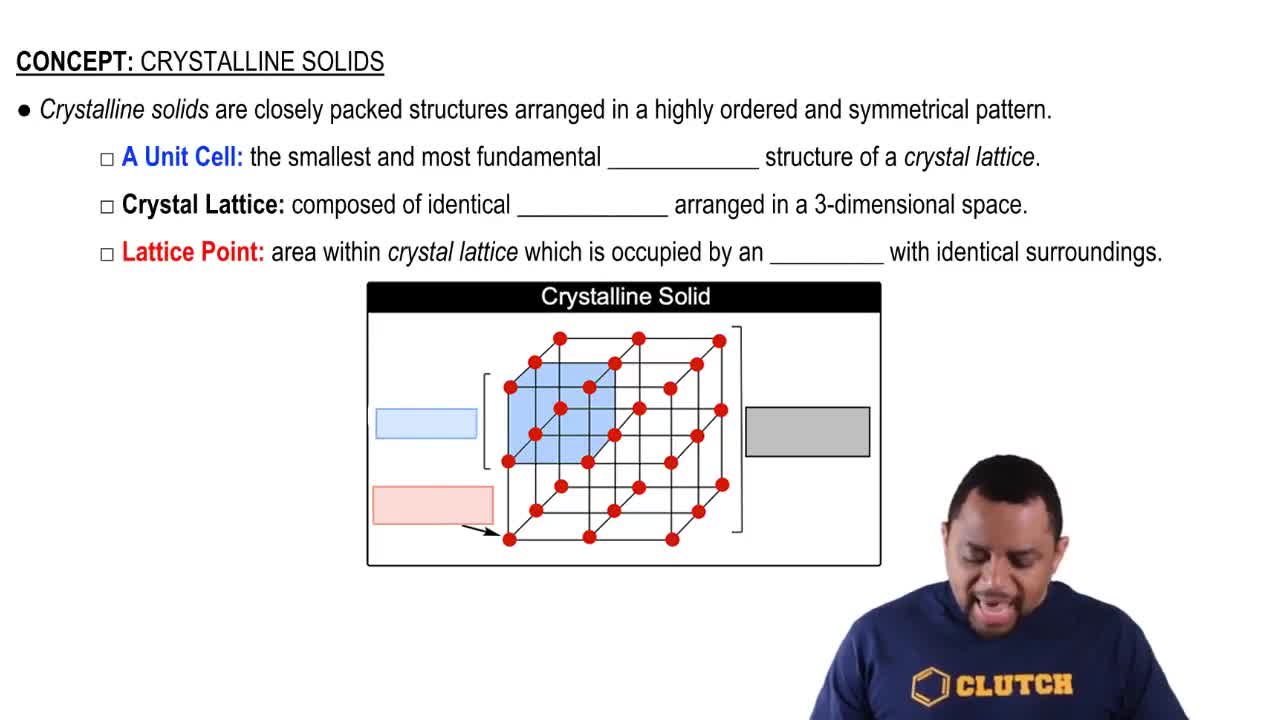
Crystalline Solids
Problem 108a
Textbook Question
Textbook QuestionWe will see in Chapter 12 that semiconductors are materials that conduct electricity better than nonmetals but not as well as metals. The only two elements in the periodic table that are technologically useful semiconductors are silicon and germanium. Integrated circuits in computer chips today are based on silicon. Compound semiconductors are also used in the electronics industry. Examples are gallium arsenide, GaAs; gallium phosphide, GaP; cadmium sulfide, CdS; and cadmium selenide, CdSe. (b) Workers in the semiconductor industry refer to 'II–VI' and 'III–V' materials, using Roman numerals. Can you identify which compound semiconductors are II–VI and which are III–V?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
291
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos