14. Solutions
Solutions: Solubility and Intermolecular Forces
Problem 99
Textbook Question
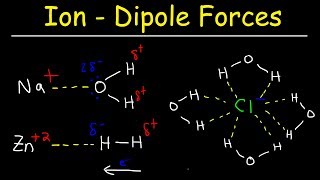
Textbook QuestionThe solubility of carbon tetrachloride (CCl4) in water at 25 °C is 1.2 g>L. The solubility of chloroform (CHCl3) at the same temperature is 10.1 g>L. Why is chloroform almost ten times more soluble in water than carbon tetrachloride?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
2741
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos