10. Periodic Properties of the Elements
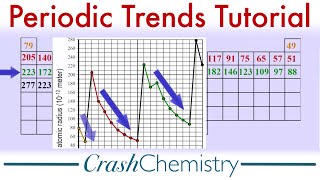
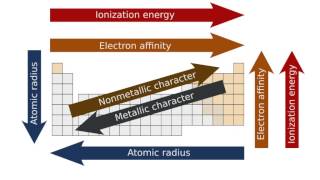
Periodic Trend: Cumulative
Multiple Choice
Multiple ChoiceWhich of the following statements is true?
A
Valence electrons are more difficult to remove than inner core electrons.
B
Argon and bromide are isoelectronic species.
C
Francium possesses the greatest electronegativity.
D
The second ionization energy of an atom is always larger than its first ionization energy.
E
The aluminum atom will experience a larger jump in its third ionization energy than its fourth.
467
views
Related Videos
Related Practice
Showing 1 of 8 videos