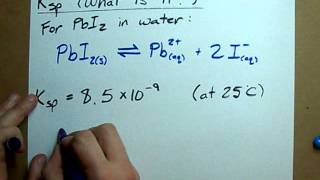18. Aqueous Equilibrium
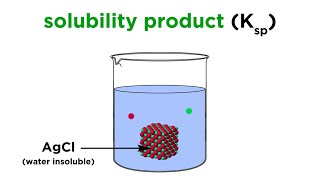
Solubility Product Constant: Ksp
Problem 150
Textbook Question
Textbook QuestionA saturated solution of an ionic salt MX exhibits an osmotic pressure of 74.4 mm Hg at 25 °C. Assuming that MX is completely dissociated in solution, what is the value of its Ksp?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
481
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos