16. Chemical Equilibrium
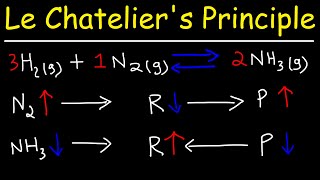
Le Chatelier's Principle
Multiple Choice
Multiple ChoiceA container is charged with 2.00 mol of I2 and is allowed to reach equilibrium. The I2 decomposes to iodine atoms as shown in the following reaction:
I2 (g) ⇆ 2 I (g)
After the reaction reaches equilibrium, an inert gas is introduced into the container under conditions of constant volume. Which of the following would you expect to happen?
A
[I2] will decrease.
B
The partial pressure of I2 will increase.
C
[I] will increase.
D
The partial pressure of I will increase.
E
No change; the reaction is at equilibrium.
210
views
Related Videos
Related Practice
Showing 1 of 13 videos