17. Acid and Base Equilibrium
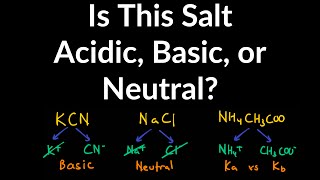
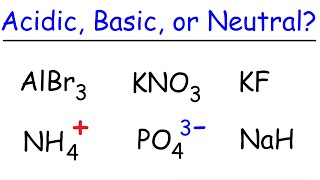
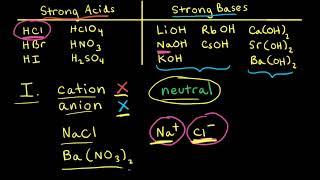
Ionic Salts
Problem 64b
Textbook Question
Textbook QuestionAn element X reacts with oxygen to form XO2 and with chlorine to form XCl4. XO2 is a white solid that melts at high temperatures (above 1000 °C). Under usual conditions, XCl4 is a colorless liquid with a boiling point of 58 °C. (a) XCl4 reacts with water to form XO2 and another product. What is the likely identity of the other product?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
710
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos