10. Periodic Properties of the Elements
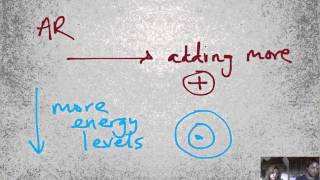
Periodic Trend: Atomic Radius
Problem 24
Textbook Question
Textbook QuestionThe experimental Pb¬Cl bond length in lead(II)chloride, PbCl2, is 244 pm. Based on this value and data in Figure 7.7, predict the atomic radius of Pb.

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
952
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos