12. Molecular Shapes & Valence Bond Theory
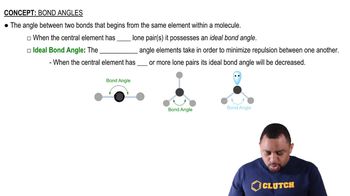
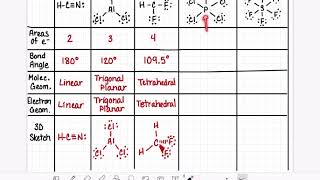
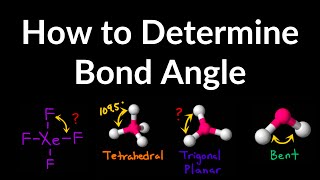
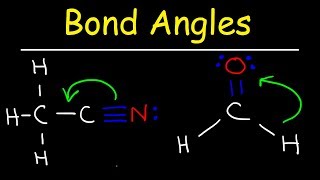
Bond Angles
Problem 115
Textbook Question
Textbook QuestionThe phosphorus trihalides 1PX32 show the following variation in the bond angle X¬P¬X: PF3, 96.3°; PCl3, 100.3°; PBr3, 101.0°; PI3, 102.0°. The trend is generally attributed to the change in the electronegativity of the halogen. (b) What is the general trend in the X¬P¬X angle as the halide electronegativity increases?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1423
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos