12. Molecular Shapes & Valence Bond Theory
Molecular Orbital Theory
Problem 112a
Textbook Question
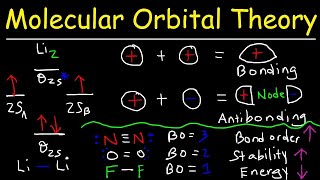
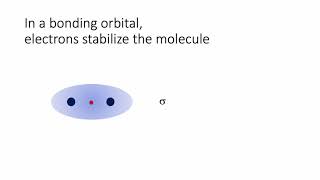
Textbook QuestionThe energy-level diagram in Figure 9.36 shows that the sideways overlap of a pair of p orbitals produces two molecular orbitals, one bonding and one antibonding. In ethylene there is a pair of electrons in the bonding π orbital between the two carbons. Absorption of a photon of the appropriate wavelength can result in promotion of one of the bonding electrons from the p2p to the p*2p molecular orbital. (b) Assuming this electronic transition corresponds to the HOMO-LUMO transition, what is the LUMO in ethylene?

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
475
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos