11. Bonding & Molecular Structure
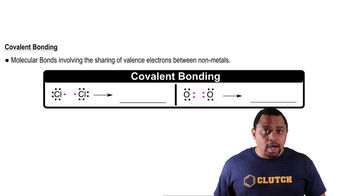
Chemical Bonds
Problem 27
Textbook Question
Textbook QuestionEnergy is required to remove two electrons from Ca to form Ca2+, and energy is required to add two electrons to O to form O2 - . Yet CaO is stable relative to the free elements. Which statement is the best explanation? (a) The lattice energy of CaO is large enough to overcome these processes. (b) CaO is a covalent compound, and these processes are irrelevant. (c) CaO has a higher molar mass than either Ca or O. (d) The enthalpy of formation of CaO is small. (e) CaO is stable to atmospheric conditions.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1081
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos