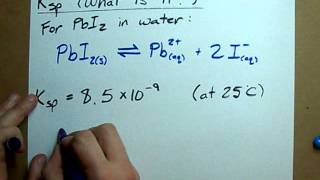18. Aqueous Equilibrium
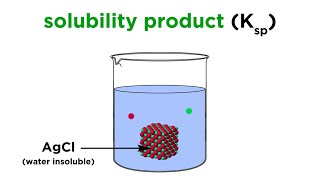
Solubility Product Constant: Ksp
Problem 117
Textbook Question
Textbook QuestionA concentration of 10–100 parts per billion (by mass) of Ag+ is an effective disinfectant in swimming pools. However, if the concentration exceeds this range, the Ag+ can cause adverse health effects. One way to maintain an appropriate concentration of Ag+ is to add a slightly soluble salt to the pool. Using Ksp values from Appendix D, calculate the equilibrium concentration of Ag+ in parts per billion that would exist in equilibrium with (c) AgI.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
935
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos